 Principles:
Principles:This is an example of so-called "rapid tests" to detect infection by coronavirus (SARS-CoV-2, causing the COVID-19 disease). In about 15 min it allows to detect in blood, serum or plasma the presence of antibodies against a protein which is part of the virus capsid.
 Principles:
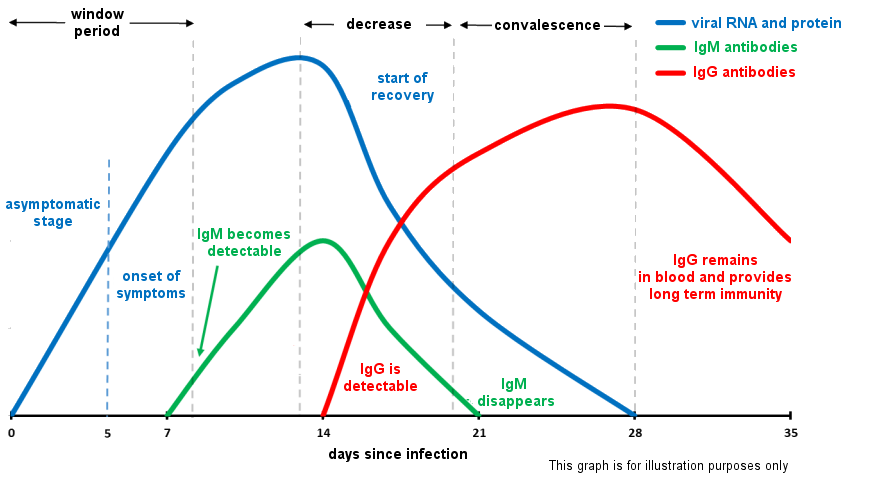
Principles:The presence of antibodies in patients' blood indicates they are, or have been, infected by the virus. Antibodies of the IgM type are the first to be produced (primary response, early response), followed after several days by antibodies of the IgG type (secondary response, delayed but of a longer permanence)
As a consequence, detection of just IgM indicates a primary infection, simultaneous detection of both IgM and IgG indicates a secondary infection; detection of just IgG indicates probability of a secondary infection (or a later stage of infection).
This technique is called immunochromatography or, also, lateral flow immunoassay. The sample is applied on an absorbent area where it gets mixed with some reagents previously deposited there, and then molecules in that mixture move by capillary effect along a strip of nitrocellulose membrane. In some areas of this strip specific antibodies have been previously fixed or immobilised, which will capture their respective antigens, or vice versa (together with whichever molecules that are bound to these).
 In the examples here displayed, some molecules are attached to colloidal gold nanoparticles, of a red colour. This provides very easy detection of the results.
In the examples here displayed, some molecules are attached to colloidal gold nanoparticles, of a red colour. This provides very easy detection of the results.
This system allows for a rapid detection, without the need for any instrumentation; a visual inspection is enough. The use and the interpretation of results are similar to those of the well known pregnancy tests.
Let's see two examples of design of these devices:
Antibodies (human) in the sample: IgG type;
IgM type (both specific for the virus);
non-specific IgG
Control protein: chcicken IgY
Reagent for detection: colloidal gold as nanospheres (~40 nm ⌀)
Antibodies as reagents: from goat against human IgG;
from goat against human IgM;
from goat against chicken IgY
Antigen as reagent: N protein, a fragment of the virus capsid (recombinant protein, obtained with genetic engineering)
With this format, the assay can only detect one type of antibodies. Two designs with this format are demonstrated next, one directed to IgG and the other to IgM.
Pick an option and then click successively on the image to the right for the animation to run step by step. If that doesn't work (mobile devices), swipe leftwards.
Items in the diagram are not drawn to scale.
If there is no line at "C", whatever is displayed at the other areas is irrelevant
Antigens immobilised at the T area will specifically capture the antibodies that might be present in the sample (IgG or IgM, depending of the design of the device); these will have bound the secondary antibody (anti-Ig) in turn linked to the gold particles, which will be detectable as a red line in the T area.
Antibodies immobilised at the C area will specifically capture the IgY used as control (previously deposited in the sample application area), which is linked to gold particles, which will be detectable as a red line in the C area. This line ensures that sample and reagents application and migration have been correct. If there is no line at C, results must not be interpreted since the assay has not worked properly (it is irrelevant whether there is or not a red line at T).
Summarising:
| (soluble) control protein | (soluble) antibody in the sample application area |
antigen immobilised at the T area | antibody immobilised at the C area | |
| Design for IgG: | ||||
| Design for IgM: |
This format allows simultaneous detection of both types of antibodies, IgG and IgM.
Antibodies (human) in the sample: IgG type;
IgM type (both specific for the virus)
Control protein: rabbit IgG
Antigen as reagent: N protein, a fragment of the virus capsid (recombinant protein, obtained with genetic engineering)
Reagent for detection: colloidal gold as nanospheres (~40 nm ⌀)
Antibodies as reagents: from goat against human IgG;
from goat against human IgM;
from goat against rabbit IgG
Pick an option and then click on the image to the right
for the animation to run in steps.
Items in the diagram are not drawn to scale.
If there is no line at "C", whatever is displayed at the other areas is irrelevant
Antigens immobilised at the M area will capture the IgM that might be present in the sample (early immune response); if these recognised the viral antigen they will also carry the gold particles, which will be detectable as a red line in the M area.
Antigens immobilised at the G area will capture the IgG that might be present in the sample (late immune response); if these recognised the viral antigen they will also carry the gold particles, which will be detectable as a red line in the G area.
Antibodies immobilised at the C area will specifically capture the IgG used as control (previously deposited in the sample application area), which is linked to gold particles, which will be detectable as a red line in the C area. This line ensures that sample and reagents application and migration have been correct. If there is no line at C, results must not be interpreted since the assay has not worked properly (it is irrelevant whether there are or not red lines at M or G).
It is possible to use other types of antibody, following the same specificity principles:
| In the schematic shown: | rabbit IgG | goat IgG against human IgM | goat IgG against human IgG | goat IgG against rabbit IgG |
| An alternative design: | mouse IgG | mouse IgG against human IgM | mouse IgG against human IgG | goat IgG against mouse IgG |
If there is anything you don't understand or you have any doubts, please That will help us improve this page.